Swords in the Bronze Age by J.M. Ney-Grimm
Tracking down knowledge is my drug of choice. Each new fact is just so interesting! Even better is the moment when an entire constellation of facts coalesces, and I see how it all fits together and what it all means. That’s a total thrill!
But my insatiable curiosity (and I seem to be able to be curious about everything and anything) was not why I researched bronze metallurgy in ancient times. I was writing a novel set in the Bronze Age of my North-lands, and my protagonist was essentially the treasurer for a warlord. The wealth of the citadel lay in its metals and – especially – its weapons. So I needed to know all about how the metals were extracted from the earth, how they were purified and poured into ingots, and what forging techniques were used. My protag knew all that stuff, so I needed to know about it also.
For those of you who share my curious bent, here’s what I discovered.
Gritty DetailsToo many of the sources I found were overly theoretical. The author might explain why ancient cultures developed metallurgy as they did or how they traded for their tin. But I needed nitty gritty details.
How were their smelting furnaces set up? How long did it take for the metal to become molten? How exactly did the ancients fashion bronze scale mail? How did they make their bronze swords?
Historical re-enactors and experimental archeologists proved to be my most fruitful sources. I found actual patterns for re-creating bronze helmets and bronze armor, along with photos of the finished results. I found videos showing Bronze Age combat techniques.
Smiths Were MagesThe website of Neil Burridge, a smith who creates Bronze Age artifacts using authentic materials and methods, had the details I was truly seeking. Videos of him in action allowed me to see a real smith moving within the forging environment, garbed in the protective gear of heavy apron and gauntlets, using the tongs and crucibles, exercising prudence with the liquid fire that is molten metal.
He also explained vividly the awe with which the ancient smiths were probably regarded. Metallurgy was not a theoretical science for them. It was a practical discipline, absolutely necessary for their tools and weapons, but with techniques developed over hundreds of years and handed down from one smith to another.
They didn’t know why these techniques worked. And they weren’t infallible. Sometimes a pour would turn out a perfect result. Other times it would fail, and the smith wouldn’t know for sure what had caused the failure. Certainly ordinary people, with no access to a smith’s secrets, would have regarded the whole business as magical.
Why Would a Skilled Smith Waste His Time?Although the people in my novel were using Bronze Age technologies, I envisioned them as possessing military organization more like the armies of ancient Rome. Thus my smiths would not spend days setting up for the pouring of one sword that might – or might not – deliver success. They would pour many blades in one day, and then hand the blades off to others for the steps that transformed the plain metal blank into a weapon.
Anvilfire.com, a website “dedicated to advancing modern blacksmithing while retaining traditional standards of craftsmanship,” supplied me with information about this finishing process. The bladesmith created the blade. A separate shop did the grinding and polishing. Yet a third made the hilt and secured the blade to it. And a fourth made the scabbard.
Making a sword was resource intensive, both because of the valuable metals required and because of the labor from many skilled individuals that went into it.
Firesetting at the Copper MineSo what about those materials? Bronze is made by mixing a small part of tin with a larger portion of copper. The ancients didn’t have modern strip mines or deep underground mines. Nor did they have sophisticated machinery run by deisel engines. How did they get copper and tin out of the ground?
Copper mines bore some resemblance to my expectations. The copper deposits needed to be relatively near the surface, but the ancients actually did tunnel down to a vein of ore. There, at the working face, they built a fire to heat the ore-containing rock. Once the rock reached a high enough temperature, they doused it with cold water. This process increased the brittleness of the rock and induced a preliminary degree of cracking. Blows from a hammer or pick could then break it into rubble, which could be heated in a smelting furnace to extract the copper.
StreamworksTin was another matter, one entirely new to me.
Tin was found in alluvial deposits in stream beds, usually as a very pure tin gravel well stirred with gravels of quartz, mica, and feldspar (gangue). So the trick was to separate out the tin gravel from the others.
The method of the ancients, as far back as 2,000 BC, was this:
• Dig a trench at the lowest end of the deposit.
• Dig a channel from the nearest water source to pour water over that part of the deposit
• Allow the stream of water to wash the lighter gangue into the trench
• Pick up the heavier tin gravel that remained
• When the lower portion of the deposit had yielded all its tin, dig another trench a bit higher and redirect the water channel, to allow the next section of the deposit to be harvested
The tin gravel thus obtained would be roughly smelted on site, simply roasting the gravel in a fire. The pebbles resulting from this rough smelt would then be transported to a dedicated furnace for a second smelting that yielded the purer tin needed by bladesmiths.

What About the Ingots?Modern ingots are rectangular blocks, but those of the ancients took several different forms. The earliest were so-called “biscuit” ingots, round on the bottom like a muffin, gently concave on the top. They took the shape of the earthen pit into which the molten metal dripped from the smelting furnace.
But metal is heavy, and the biscuit shape awkward to carry. Around our own Mediterranean, an “oxhide” form was developed. It weighed about 80 pounds and possessed four “legs,” one at each corner, that allowed it to be tied between pack animals or gripped and carried by men.
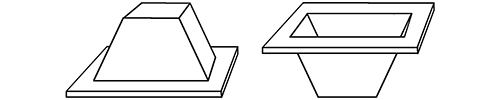
I became fascinated with an ingot form used much later by the Chinese in the Malay Penninsula. These were hat shaped, much smaller (weighing only a pound), and actually used as currency.
A Peculiarity of Forging in BronzeBronze has one very peculiar property in the smithy.
Most metals, such as iron or even copper, when heated and cooled slowly to room temperature, become more ductile and more workable. They are less prone to internal stresses.
Bronze does not behave like this. When slow cooled, it becomes brittle and difficult to work. Thus it must be heated to cherry-red and then quenched in water. This quick cooling makes it so soft that it can then be hammered. The hammering condenses the metal, giving it more rigidity.
A bladesmith will hammer near the edge of a blade to harden it and help it keep its sharpness, while allowing the center rib to retain more of its resilience.
Were These Swords Any Good?If you compare a bronze sword to a steel sword, the steel is always going to win. But when the Bronze Age gave way to the Iron Age, bronze metallurgy was at its peak. Several thousand years had gone into the development of the most superb techniques. Iron metallurgy was in its infancy, and getting the iron swords to be rigid enough was a problem. The iron swords just weren’t as good as the bronze ones, which were light, strong, just rigid enough, and held an edge well.
But there’s no need to take my word on this. A YouTuber with a passion for swords, Skallagrim from Canada, discourses quite knowledgeably about the pros and cons of bronze. More amusingly, he tests one of Neil Burridge’s bronze swords “to destruction” in the video below.
(There’s a brief reprise snippet of Mr. Burridge before Skallagrim gets going with his destruction. Go to the 3:30 mark, if you want to skip that snippet.)
Even after all my research, I cannot call myself more than a mere smatterer. I learned enough to write The Tally Master, and not much more. But I hope you found these tidbits entertaining, and I’ll be happy to answer questions in the comments below. Or to speculate with you, when I don’t know the answer. ;)
Whoo hoo! Neato!
Now I shall go back and read this entirely through. Thank you.
LikeLike
Thank you for sharing all that. It was fascinating!
LikeLike
Agreed.
LikeLike
Yes, thanks! I have a personal interest in the neolithic and bronze age and didn’t know a lot of this or know about Neil Burrage’s site. Or about the hat-shaped ingots.
What is my personal interest? Why I’m glad you asked because I can talk about it until everyone finds an excuse to be somewhere else! I have a world-building hobby but don’t follow up with the stories part.
Anyway, in one of my settings space aliens kidnap humans (and various plants and animals) from a few bronze age civilizations, modify them a bit, and put them on an exomoon with some other aliens. Unfortunately, traditional metallurgy doesn’t work due to the local atmosphere (higher partial pressure of oxygen, lower percent of oxygen). So almost everyone’s stuck in the stone age.
They have polymer mirrors (from a ‘tree’), so in theory they could make a large enough solar furnace to smelt ore, but only one ‘nation’ of one kind of alien has done that yet (and they’re far away and communicate by excreting ‘silk’ with chemical signals, so good luck explaining that you want bronze in a shape useful for humans).
LikeLike
Cool! I find the world building that I do before and during a story to be a lot of fun, so I understand the fascination of your hobby. Thanks for sharing your space alien-kidnapped humans-exomoon milieu. Neat stuff!
LikeLike
One of the tricky aspects is that tin is not a common metal. The geological processes that concentrate tin compounds into ores, especially ores workable with Bronze Age technology, are unusual. Hence you get things like tin being shipped from the British Isles over large parts of Europe. Some of the sources I was able to track down claimed that tin was the third most precious ancient metal, after silver and gold.
LikeLike
Exactly! It surprised me to learn just how far certain rare materials, such as tin and amber, traveled during the Bronze Age.
LikeLike
amber is just for jewelry?
LikeLike
Yes, jewelry, but it was also used in the medicinal remedies of the time.
Pliny makes a reference to its use in drop spindles for its electrostatic properties.
LikeLike
I had never seen a reference to amber in spindles before, that is interesting. Static charge can be a real issue when dealing with spinning fiber.
LikeLike
Smiths Were Mages
Michael Scott Rohan, who had “Mage-Smiths” in his Winter Of The World series, talked about the historical beliefs that link Smiths & Magic.
Of course, Rohan’s main character was somewhat based on Wayland/Völundr.
https://en.wikipedia.org/wiki/Wayland_the_Smith
LikeLike
Interesting stuff! Thanks for sharing the link!
LikeLike
Wizarding used to require actual work back then.
LikeLike
In the Lorica — the Breastplate of Saint Patrick — there is an invocation against “spells of witches and smiths and wizards,”
LikeLike
One of the things I liked about The Treachery of Beautiful Things by Ruth Frances Long was the scene where Jack had to get his sword from a legendary smith.
LikeLike
Quick technical point – not all bronzes used tin. There were copper-arsenic and other blends that are considered “bronze” and that were used for jewelry and tools. They have different qualities from tin-copper bronze, but were relatively common, especially in Central Eurasia (Black Sea to China).
LikeLike
Excellent point. Especially since the ancients were, to a large degree, at the mercy of what came out of the ground. Different ores possessed different mixes of impurities. Thanks, TXRed!
LikeLike
Copper-arsenic? There have to be some interesting legends/folklore about the smiths who worked with that material.
I’m thinking along the lines of “Mad as a hatter.”
LikeLike
Oh, yeah! Apparently it affected the eyes, lungs, and skin of smiths, and damaged the nerves, often causing lameness.
https://en.wikipedia.org/wiki/Arsenical_bronze#Health_effects_of_arsenical_bronze_use
LikeLike
Probably where we get the legend of smiths being lamed by the gods so they wouldn’t flee?
LikeLike
Possibly why even divine smiths such as Hephaestus were lame.
LikeLike
Strikes me as more probable that lame men were put to the task of smithing, since most other strength-requiring things also require walking and running.
LikeLike
Probably a little from column A, and a little from column B.
LikeLike
Would YOU trust a guy whom you had deliberately mutilated to make the sword you needed in battle?
LikeLike
Maybe if I was the ruler and I knew that my heirs would kill him painfully if my sword failed. :evil:
LikeLike
And many an heir has counted on that reasoning…. ;-)
LikeLike
Didn’t say that it was “completely smart”. :evil:
LikeLike
Oh, if you read my Wiki link (above) concerning Wayland/Völundr, you could see why it wasn’t very smart.
IE look at what Völundr did about his enslavement and being crippled. :evil: :evil: :evil: :evil:
LikeLike
That would require their knowing it was the sword, and that it was your fault. Swords broke, after all. Who’s going to waste a smith so valuable that you mutilated him to keep him from running away when it might have been mere chance?
LikeLike
Point.
Although I wonder if the story of Wayland/Völundr got started by smiths to warn about “what might happen if that happened to a smith”. :twisted:
LikeLike
If all the swords from Joe Smith seem to fail, people might get suspicious.
LikeLike
Joe’s got to use judgment. Either take out the worst, or make all their swords fail at once, so they can’t come back and do something about it.
LikeLike
Getting multiple fails simultaneously would seem like magic.
LikeLike
Evidently chronic arsenic poisoning results in peripheral neuropathy which often leads to leads to weakness in the legs and feet. Thus it is likely that smiths working continuously with arsenical bronze developed neuropathy and concomitant lameness, and the cultures surrounding these lame smiths envisioned their smith-gods as lame like their smiths. More of the retconning you mention downthread.
LikeLike
Could also explain why Hephaestus was described as lame.
LikeLike
Likely the reason he was originally said to be lame, but the later myths have Hephaestus as a child thrown from Mount Olympus and was lamed by the fall. :wink:
Apparently, Hera (his mother) thought him to be an ugly child. :grin:
LikeLike
Ret-conning things like that is common. Like the Areopagus being the place where the first trial was of Ares. (Poseiden prosecuted him for killing his son; Ares’s defense was, depending on the version, that the son was trying to, or had, raped his daughter; he was acquitted.) which was certainly invented to explain the name.
LikeLike
Haven’t found too much on the ill-effects of working that, but there is some about the arsenical-gold favored by the nobility of Chan Chan (Moche culture) in what is now Peru. Yes, it was a little rough on the smiths, especially since they used blow-pipe furnaces to melt the ores. Inhaling arsenic vapors….
I’m afraid trying to find English-language material on the Central Asian sites is areal challenge. Russian-language has a lot richer selection, IF you can find an institution that has the journals and monographs.
LikeLike
The Moche are fairly high on the “Please don’t time travel there” scale. I mean, anybody can make a skull cup. The Moche made skull banquet sets.
LikeLike
It makes lovely dyes. It’s at least a suspect in the tales of clothes dyed in dragon’s blood that are so lethal, though myth-making would speed up the process.
LikeLike
Thank you, thank you, thank you!!
LikeLike
I have needed those exact same sorts of details so many times. Thank you for this!
If bronze was at its heyday when iron began to be smelted, now I wonder if the transition didn’t have more to do with difficulties in making bronze than with the superiority of iron?
LikeLike
IIRC to a degree yes.
Iron was commoner than tin so Iron tools became more common than bronze tools.
One fictional character commented that all of the men of an iron-forging tribe could be armed with iron swords but bronze swords were too expensive for another tribe to arm all of their men.
LikeLiked by 1 person
Aristotle noted the difference that made to political structures.
LikeLike
My understanding is that the switch was due to the rarity of tin. There wasn’t enough of the stuff, readily available, to produce the arms and armor needed to equip the larger armies that were becoming prevalent. Better to have every fighter equipped with iron gear than to have him unarmed and awaiting the completion of a bronze sword.
LikeLiked by 1 person
This is why Portugal and the British isles had a trading relationship all the way to pre-history. Portugal (in the North) has vast deposits of tin.
LikeLike
Gee, that gives a whole new perspective on the phrase “He’s got a tin ear.”
LikeLike
Is Portugal where Rome got its tin from? Is tin the reason for the Roman colony in Britannia? I mean today’s GB not Brittany in France.
LikeLike
I thought there was tin in Cornwall. Was it not discovered until much later?
LikeLike
I have no idea! I know this goes back tot he 4th century bc, because of some stuff about it on (of all things) a program on Stonehenge.
LikeLike
The Phoenicians were trading with Cornwall and Wales for tin in the 4th C BC, evidenced by the distinctive “hide-shaped” tin ingots from Southern Britain found in the Eastern Med in Phoenician shipwrecks, and by a Phoenician bronze sword found in a 4th C BC grave in Cornwall.
To me, the standardized shape / size (appx. 80 Lb) of the hide-shaped ingots suggests a regular and developed trade route with reliable trading partners.
As tin is the smaller fraction of bronze, it could be economically imported into a copper-rich area, as was done by several groups of Celts, like the Luristan culture and the Galatians near Ankara, the earlier Hallstatt towns in Austria, and the Celts of the Northwest of the Iberian peninsula, all of whom were settled on trade routes in places where salt and copper were found. The NW of Iberia was also a source of tin.
This is a rich and interesting area to research, and many people have done so, even in English. A book by Georgius Agricola, De Re Metallica, is a cheap and accessible book on Medieval metalworking, smelting, and casting, and is a good starting point for further research.
JPDev
LikeLike
I just found this article about sources of tin in ancient times. Pretty interesting stuff!
https://en.wikipedia.org/wiki/Tin_sources_and_trade_in_ancient_times
LikeLike
My recollection (of an ’80s-era documentary) is that the tin trade was disrupted, forcing consumers to use recycle (or resort to copper and/or soft iron), and later the Assyrians started producing “good iron” (aka steel), which ushered in the Iron Age.
LikeLike
Oh, nice detail! Thanks.
LikeLike
Steel was made as far back as the Assyrians? Wow!
LikeLike
There are different stages in making steel. It’s easy to get carbon into the surface of a mass of iron, and thus have an outer layer of steel over a core of wrought iron. At a more advanced stage you can heat the iron enough to make it bendable, fold it over, and hammer it flat, producing layers with different amounts of carbon. If you do this lots of times you get many layers of steel, approximating to solid steel in a fashion that you probably need calculus to describe; I believe this is how the Japanese “folded metal” technique for making swords worked. Then eventually you a furnace hot enough to completely melt the iron so that it can be stirred; I think that was later medieval. Finally, you have the techniques of the Industrial Revolution that made good steel cheap to produce in mass quantities.
If you want a beautiful metallurgical scene, take a look at the second chapter of Atlas Shrugged, which starts with the first heat of Rearden Metal (described as a new alloy that is to steel what steel is to iron). It’s not far short of erotic.
LikeLike
Damascus steel was also a folded metal thing. But the Cistercians made real steel in factories, which was why the French king gradually took away all their lands and factories for himself.
LikeLike
I think it was also the Cistercians who were working on the experimental blast furnace in Northern England, right before Henry VIII grabbed their monastery and turned out the experimenters.
LikeLike
Depending on how you make your iron, the problem can be to remove the excess carbon (at least with 19th century and onward processes). Most steel has, oh, less than 1/2% carbon by weight. Cast iron, wrought iron starts, IIRC, 2 to 3 percent by weight carbon.
LikeLike
As i learned the terminology, cast and wrought iron are different substances: Cast iron has high carbon content, wrought iron low.
LikeLike
Correct, steels end at 2%C by weight, then the cast irons begin (and your carbon content becomes too high for the heat treating of steel. Wrought iron is more a processing category than a composition range.
LikeLike
A large issue was the significantly higher melting temperature of iron, thus making it significantly more difficult to refine into steel or otherwise heat treat with the technology of the time. While ultimately superior, different techniques were required.
LikeLike
Looks like you don’t have to melt to get iron from ore. The bloomery process (see https://infogalactic.com/info/Bloomery) uses a reduction process to get pure-ish iron. The bloom needs to be wacked with a hammer to get rid of the rocky slag, but you don’t need near as high a temperature. With a lot of care, you can get some nicely forgeable iron.
I have a bunch of magnetic dust around (downwind of Crater Lake), and I’ve wondered if iron could be made.
LikeLike
That is true, but the bloomery process often created more cast iron than steel (cast iron has more than 2% by weight carbon). Then subsequent processes make it useful.
The magnetic dust would be smeltable, depending on the content. If it’s magnetic, you’re looking at ferritic iron, cobalt or nickel.
LikeLike
There was also some pure meteoric iron. This features in the Bronze Age.
LikeLike
There’s this guy, who, in one of his videos, was able to get iron and make a small ingot. It used an interesting source but my net is too crap for me to open it now.
https://www.youtube.com/channel/UCAL3JXZSzSm8AlZyD3nQdBA/videos
Actually, I find most of his videos fascinating to watch. I figure the folks here will too!
LikeLike
Oh, yeah! Love the Primitive Technology videos. Fascinating, indeed.
LikeLiked by 1 person
I am just going to bookmark this entire discussion. It’s got so many good resources in it.
LikeLike
You can read the history of human technological progress as a succession of resource crises. The first was probably when they ran out of easily available flint in some regions, which was replaced by bronze, which was replaced by iron when the tin became harder to get, which… You get the point.
Same stuff, different era: Charcoal running out? OMG… Whatever will we do…? Oh, wait: Coal. Coal starts to have issues…? Oh, wait, here’s oil; easily recoverable oil runs shy? Cue the discovery and use of shale oil and other fracking-based recoverable hydrocarbons.
Resource crises are generally frauds; as soon as we run out of something, a replacement is found. No doubt, there will continue to be such things going forward, and we’ll simply find substitutes, or move on to better and more efficient resources.
LikeLiked by 1 person
Turns out that in some cultures, bronze and stone were kept as status and ritual implements even after iron appeared. So you can find all three in some of the Iron Age kurgan burials in Central Asia.
LikeLiked by 1 person
I’ve been occasionally rooting around picking bog iron nodules out of our swamp for the past several years. Only have a couple of cups worth, not really enough to try smelting them yet. So I either need to keep doing it for a bunch more years at current rate, or get serious about harvesting.
LikeLiked by 1 person
That youtube of the bronze sword was -very- impressive. I learned a lot from that.
LikeLiked by 1 person
c4c
LikeLike
D5D
LikeLike
c4c2
LikeLike
My goodness, that was interesting. Thanks for repurposing your research.
LikeLike
Buddy of mine and I recently looked at making a bronze sword. Then we looked at the price of tin. Nope. Secondary solution was pennies (one era was practically bronze in it’s own right). Wasn’t going to happen. Materials cost was just too impractical.
I will be going over these videos sometime this weekend when I have time. Just because I am an information junkie. :)
LikeLike
I pay about $25-$30 per pound for the 99+% tin I buy for making bullet alloy.
My inner Scotsman twitches a bit every time some goes downrange, but it’s still cheaper than jacketed bullets…
LikeLike
Tin is more expensive than copper? Hmm….checking onlinemetals.com: 2.25 inch diameter bronze 544 round, random lengthen (10 to 12 inches): $198.62 (meets ASTM B139/B139M). Okay, not cheap; not the commodities market either; it’s hard to find a listing for tin.s
LikeLike
Ouch; north of $20,000 per ton (US/US).
LikeLike
For making a sword, you would not need very much tin, about 10% by weight, and as you can cast bronze nearly to finished shape, you should not need more than 7 or 8 Lbs of bronze in your crucible. I generally use the 95/5 lead-free solder as my tin source, and used copper plumbing pipe for my copper. You would need appx. 1 Lb (2 rolls from Lowes) ~ $ 12.
I melt in a graphite crucible appx 5″ x 10″, set in a charcoal fire, using an oxy-propane torch with a rosebud tip for direct heating of the metal. (crucible from Rio Grande Jewelry @ riogrande dot com).
Melt the copper first, clear the surface of impurities and slag with borax, then, when it is fully liquid and the mold prepared, add the tin and stir thoroughly, then cast in a quick, smooth pour until the mold is completely full.
Use appropriate protective gear, shoes, and natural fiber clothing only.
There are several good reference books on metal casting – read them.
The mold frames can be made of plywood glued up to 1.25″ to 1.75″ thick (for each mold half) with dowel pins in one side and corresponding holes in the other for registration of the mold. (This is for sand casting). The sword can be made from a wooden positive model (a soft wood like poplar or basswood is strong enough and easy to work), which is then pressed into the bonded sand in the un-pinned 1/2 of the molds, the the pinned half is set on top and more sand pressed (rammed) in. The molds are separated, the model removed, and an in-gate carved in the sane to allow the metal into the void. I often cut several grooves in the mold down from the point and away from any hand-guard or pommel overhangs, to ensure that these areas fill when poured. casting sand can be bought ready-to-go, or plain, for use with epoxy bonding agents.
This kind of work can be a lot of fun.
JPDev
LikeLike
From the above sources, weren’t bronze-age swords just a tad over a pound to about 1 3/4 pounds? Is that much needed to deal with shrinkage during cooling? Note: I know absolutely nothing about casting.
Recall a bronze axe found inside its mold in SE Asia, maybe Cambodia. IIRC, the mold was carved from stone. De we take from this they weren’t using something like green sand to cast?
LikeLike
So a planet with more accessible tin might have never left the bronze age? Or might have left it fairly late?
LikeLike
More accessible and more abundantly so. I’ve wondered about that, too. It would make an interesting basis for an alternate history tale.
LikeLike
You’d want to look at the cosmic abundance of tin, and also about the geophysical processes that result in tin ores bubbling up from the mantle. Apparently they’re rather unusual and don’t happen in a lot of places. I’m not sure how much you can boost the amount of tin.
If I wanted to rationalize it, I would look at having a planet with a higher level of tectonic activity generally, and therefore more deposits of all sorts of metals. Of course it would also have more earthquakes and volcanoes and possibly be broken up into more plates. That could be exciting! I’m not sure how you’d get more tectonic activity; I think you’d want more heat flow from the interior, which means either more radioisotopes (perhaps more heavy elements overall) or more rapid freezing out of the inner core (a lot of Earth’s thermal budget comes from the heat of crystallization of core material).
LikeLike
Oh! I love the idea of a more tectonically active world with more earthquakes and volcanoes (along with more tin). I’m having all sorts or story ideas for an archipelago area in one of my fantasy milieus. Thank you for sharing that! :D
LikeLike
Be sure to do some serious reading about planetary geophysics; I’m going off impressions from a few books and some Web searches, and I may be wrong. You might look under the heading of “economic geology.”
And you’re welcome. I was doing worldbuilding for an RPG campaign, and I wanted to start out with plate tectonics and work forward. . . .
LikeLike
Understood.
LikeLike
Great post. However, as an avid slinger, I do have to object to the first video (“Bronze Age Spear Fighting”) that claims all you do with a sling is throw it. In fact, the sling has a looong learning curve, almost as long as the atlatl. Loades is correct when he says it is deadly, especially when many slingers loose at once.
LikeLike
Thanks for the correction. I did say I was a mere smatterer, alas. ;)
LikeLike
It might also be considered that up close and personal a sling makes a decent bludgeon. Not as good as a sockful of sand, perhaps, but better than an empty hand.
LikeLike
Even in Roman times (IIRC), there were still commanders who preferred allied troops of slingers to archers. Slingers were deadly and accurate, with cheap, light weapons components available to the ordinary person. Practice space was problematic and it took a while to learn, which is probably why a lot of kid herders in wilderness pasture areas were good at it.
LikeLike
I got reasonably proficient with the atl-atl with little effort. I never mastered the sling. I was surprised at the damage I could do to my surroundings with the sling, though. I just never learned to hit any *specific* surrounding…
LikeLike
It’s been years, and I couldn’t hit bulls’ eyes with the thing, but try stepping forward with the foot of the same hand you’re using to sling. Or was it the other foot? It’s been a long time, and with no one to show me how, I twirled it over my head before letting go.
DWIW, for a modern slingshot, I practiced on getting the projectile in line with the target, then practiced elevation until I hit the target.
LikeLike
Very Good!
LikeLike
Thank you.
LikeLike
I will watch the videos and read the comments later, but thanks for posting this. My son (high school freshman) got me hooked on Forged in Fire. He’ll love it when I show this to him.
LikeLike
See History Channel, Tuesday nights.
LikeLike
Yes, yes, completely off-topic, but I hardly know what I’m doing on computers and it was absolutely IMPERATIVE that I get this in front of our mighty Space Princess, and as many of you as possible, immediately! BECAUSE IT’S THE GREATEST CAT VIDEO EVER. (Please tell me if it didn’t go through.)
LikeLiked by 1 person
GUILTY!
LikeLike
Not entirely off top, since cats use swords … err, carry knives.
LikeLike
Nothing to see here. Go about your business . . .
LikeLike
“This drawer wasn’t open, and I was never here…”
LikeLike
As I recall from my own metallurgical experience (let me tell you ALL THE DIFFERENCES between 312 and 314 stainless, if you have trouble with insomnia…) the big problem with bronze is, as you allude in the forging detail, brittleness. I suspect a lot of the legends involving “sons reforging the shattered sword of their fallen father” was due to a) sword shattering in battle and b) expense of bronze :D Iron, especially at the beginning, did have issues that were gradually overcome just like with bronze, but it is more ductile and really, really hard to shatter without something like gunpowder or huge amounts of steam pressure.
I also think iron ore was prone to picking up trace elements that affected the properties (see 312/314 above…) so it wasn’t necessarily that “Toledo steel” was only good because of the smiths, but because of the local trace impurities in their ore source.
LikeLike
So there might be people who specialize in the history of metallurgy?
LikeLike
There are people who are big enthusiasts. One of my co-authors on GURPS Low-Tech (the latest edition) can go on and on about metallurgy.
LikeLike
Ooooohhhh yes. Just don’t get between two of them at an academic meeting unless you are really, really, profoundly, deeply fascinated by the topic.
Signed,
The Cornered Grad Student
LikeLike
As a materials engineer myself, we learn history as part of our program. The rest I just find personally fascinating.
LikeLike
My dad was a draftsman at Ryerson Steel (later INRYCO), and was fascinated by the history of technology, and technology altogether. I was trained as an EE (semiconductors, mostly), but I caught the enthusiasms too.
I had to argue with my advisor in high school. He couldn’t understand why a college-bound, Dean’s list (frequently) student wanted to take Drafting and Metal Shop. Never had any trouble with the greaser types; I knew the dirty jokes, too.
LikeLike
I recall years ago some damned author dropping into a novel the idea that the magic blades — Excalibur, Durandal, Cortana, Hauteclere — were simply blades forged from particularly well-tainted iron.
LikeLike
Yes, please, it’s been awhile since I’ve opened my copy of Smith’s “Physical Metallurgy.” :)
LikeLike
Sabrina, I worked in a steel producing factory for 35 years, and we made 316 stainless steel by the millions of pounds each year. I bet we could have fun discussions about this topic, but would bore everyone else to tears. Much of this went into Mercury Marine boat propellers, and also many of the jet skis propulsion systems. Also, some went into the food service industry along with 304 stainless.
LikeLike
*points* See? I didn’t make it up! :D Yep, I had to keep an eye on every component that went into the experimental apparatus. I did electron spectroscopy, so I was keenly interested in making sure nothing inside the vacuum chamber was prone to becoming magnetized. Austenitc stainless formulations for me!
LikeLike
Oh, it has been awhile. I thought austenitic was magnetic; it’s the one that body centered cubic or do I need to go back to Smith.
LikeLike
Ferrite is BCC, thus magnetic. So is delta iron, but it’s only stable at high temps. Austenite is FCC, thus non-magnetic. Cemetite (the second phase in most steels) does have a magnetic transition as well.
LikeLike
Thanks.
LikeLike
All the little things that differentiate the different CRES’s can be mind numbing. In a former job the base materials were 304/316 but we usually ended up with L for standard stuff for cold working properties and sometimes even LN to try and avoid some magnetic stuff (Al too soft, Ti too much).
LikeLike
… *looks around* Not sure if it WOULD bore people here to tears, just sayin’.
LikeLike
I’m fascinated by industrial chemistry, but I’m seriously Odd.
LikeLiked by 1 person
I wonder if the little factoid about if raining diamonds on Saturn is true, and random bits of ‘oooh’ will send me trying to find out more…
I’d like longevity, if only for the research capability!
LikeLike
I had a friend who once jokingly assured me that in heaven there would be a place to study assigned to me in the research library. I can only imagine. ;-)
LikeLiked by 2 people
I think a massive library would be heaven to a number of us here. 😆
LikeLike
C. S. Lewis once did a vignette about how you’d still have your personal library in Heaven–but only the volumes you’d loaned out. You get them all back, and all the thumbprints, pencil marks, dogears, etc., will have become illumination and gold leaf accents.
LikeLiked by 1 person
Oh! Thank you for sharing that. I’d not encountered that Lewis vignette, and it’s beautiful.
LikeLike
I think we have a bunch of wannabe (and a couple of actual) polymaths running around here, so not so much with the bored to tears.
*starring at Lazarus Long, tapping foot.*
LikeLiked by 2 people
There is a certain youtube weapons/tactics expert that started talking about firearms in warfare and had so obviously never handled a firearm in his life that he subsequently cast every word of his expertise into doubt for me.
Not the same person but I’ve seen Skallagrim say a few things that are contraindicated by historical or archaeological evidence, too.
LikeLike
The overlap (or lack thereof) between different spheres of knowledge can generate some strange gaps or inaccuracies in what the so-called experts say.
Some of the reenactors seem to know a lot about actual fighting with the weapons of antiquity, but not about the original creation methods. Modern smiths can create authentic replicas, but use modern methods. Experiential archeologists know about authentic creation methods, but don’t understand how the weapons were used and handled. Historians know about the social and/or technical milieu that influenced the development of ancient metallurgy. The enthusiasts (which is where I’d place Skallagrim) know bits and pieces from all of the above.
LikeLike
Many years ago I picked up a book on experimental archaeology. In the chapter on arms and armor, I read the authors’ statement that bronze had little value as an armor material. Then I reread it closely and realized that the experiment they described had not been able to obtain bronze (possibly because it was expensive?) and had tested copper as a substitute for bronze! That kind of destroyed my confidence in that particular book.
LikeLike
No kidding!
The experiential archeologists who I found most helpful for the novel I wrote before The Tally Master were the ones who made videos of their doings.
There was a whole series showing the building of a sewn-plank boat using Bronze Age tools and materials. Another showed the rowing and maneuvering of a trireme. And yet another series showed weaving on a variety of looms, the ground loom and the vertical loom being the two I needed.
LikeLike
I’ve read two different books connected with the Trireme Trust’s construction of Olympias, and found them fascinating. I also have McGrail’s Boats of the World on my shelves. I tend to prefer books or static Web pages to videos; I don’t process visual information well, and it tends to go by too quickly for me. Though still pictures can be a big help (Boats of the World is beautifully illustrated).
The bibliography for GURP Low-Tech is available through a link at http://www.sjgames.com/gurps/books/Low-Tech/ and may suggest some worthwhile reading, if you have access to a decent library.
LikeLike
Oh, very nice. Bookmarked it!
William, it’s interesting to me that you participate in the gaming world, both as a GM and as an author, because I used to be part of that world myself. I worked for Iron Crown Enterprises (ICE) from 1984 – 2000. I started off in the production department, drawing maps and floor plans and pasting galleys onto boards with wax. Then I moved into editing, editing the Narnia-based solo adventure books for a short while, and then moving on to the Middle-earth game modules. Eventually I became the art director, although I kept the editing duties. It was a fun ride.
Anyway, hello fellow gamer! ;)
LikeLike
I started playing Dungeons & Dragons in 1976, but my gaming group switched to Rolemaster soon after I joined them, and when I GMed it was with Rolemaster.
While I was at ICE, I wrote “Dawn Comes Early” and “Over the Misty Mountains Cold” in the Lord of the Rings Adventure Game system, and Gethaena in the ShadowWorld series. All long out of print these days. ;)
LikeLike
And if they were ‘in print’, DrivethruRPG would be charging original cover price for the PDF and you wouldn’t get anything.
I’m just sayin.
LikeLike
Oh, the first two were works for hire, and the third I sold outright for a flat fee. So, even were they in print, no money would flow to me in royalties.
LikeLike
I know how it works, I’m just a little ticked that the RPG company i did work for thinks they can charge full hardcover price for PDFs they they should have recovered the costs on 20 years ago.
LikeLike
C’mon now, you know that price is charged according to what the market will bear, not on recovery of invested costs.
That said, at some point a smart vendor will recognize the benefits of increased volume.
LikeLike
From the complaints and financial woes of the companies still operating, I think its more than the market will bear. I’ve also seen them charging that kind of price for *new* books, and acting like they have the same publication costs as they did for a hardcover 20 years ago.
LikeLike
I didn’t claim they’d priced it correctly, simply that price should not be based on production costs. I note that in the long term, prices below production costs equal bankruptcy (or tax write-off) and that excessive profit margins will erode.
LikeLike
I started out playing D&D at almost exactly the same time; then a little Traveller, Superhero 2044, and Villains and Vigilantes. I dropped out for a year or so when AD&D came out, and then took up RuneQuest 2. For a while I worked at writing my own systems to fit my own ideas. But about a quarter century ago, I decided that rather than work on homegrown systems, I would play in published systems and put my effort into worldbuilding and scenario creation and GMing. Around the turn of the century I started selling to Steve Jackson Games. It’s always been a secondary income source, but it’s a hobby that much more than pays for itself.
SJ Games is pretty good about economics. Most of the GURPS material they publish now is PDF only, and sells for fairly low prices, and authors get a higher percentage of that as royalties, reflecting the lack of printing costs. And their older books, the third edition of GURPS, sell at substantial discounts. GURPS is a fairly marginal seller for them, by and large, though they appear to be trying to raise its profile with the Dungeon Fantasy version, now at the printer. But they give good support.
I’m the type of GM who runs lots of different systems; I’ve run several campaigns specifically because I wanted to take a new system out for a test drive. I’ve settled on three generic systems as my standards—Big Eyes Small Mouth, second edition (now out of print), FUDGE, and GURPS; but I’ve run more GURPS than any other system, and nearly more than ALL other systems. Both my current campaigns are GURPS, for example.
LikeLike
Hell, I have the book for Theatrix….
LikeLike
Wasn’t Theatrix Asterix’s village drama queen?
LikeLike
no, it was a diceless RPG
LikeLike
The longer I worked in the industry, the less I played the games I helped produce. I gather that was fairly typical. ;) But all the skills I learned in the publishing of games have come in very handy for indie publishing my fiction.
Good for you that you remain in the thick of it.
LikeLike
I’ve never been in the publishing end of things. I’ve been a writer, and happily Steve Jackson Games is extremely supportive of their writers. And I’ve been able to run or play in an average of three sessions a month for quite a few years now, though it’s fallen off since we moved from San Diego to Riverside.
LikeLike
*waves to you from SGV*
LikeLike
Excellent!
LikeLike
I stopped watching Skal after his pronouncements-from-ignorance about the horrors of the United States. And IIRC, he’s originally from Germany (by way of Norway).
LikeLike
more reason to skip him
LikeLike
Skallagrim seems to be pretty up-to-snuff on edged weapon stuff, but then his yammerings about US politics from a smug Euro-weenie background are frequently off-putting.
Matt Easton of Schola Gladiatoria, based in the UK, is much more watchable for that reason (Easton seems to eschew political discussions).
LikeLike
I just bookmarked Schola Gladiatoria. Thanks for the rec!
LikeLike
> If you compare a bronze sword to a steel sword,
> the steel is always going to win.
I don’t want to nitpick a good article, but “bronze” and “steel” cover a huge amount of ground. Copper and tin are relatively easy to purify for practical use. There are other elements that go into types of bronze, some hard to get, some available as impurities, that substantially change the strength and hardness of the final product.
Iron, on the other hand, is simply nuts. Iron has a crystalline structure, and its characteristics vary wildly not only with different alloying elements, but by the process by which the final product is heated, forged, cooled, and combinations thereof. “Steel” is just iron with some carbon in it. The final product can be anywhere from “about as soft as pine” to “brittle like glass.”
Classic smiths and industry relied on iron from specific sources, like Sweden or the Ruhr, where the ore had the right type of impurities to result in a superior product. It wasn’t until the 1800s when chemists started to get a handle on *why*, and ferrous metallurgy still had a lot of “Heere Be Draggons” areas up into the 1950s.
A good steel sword might be better than a bronze sword… but a bad steel sword would be a lot worse.
LikeLike
Excellent points all.
LikeLike
Copper (and thus bronze and brass) and all metals are crystalline. Amusingly, the ‘crystals’ new agers love aren’t crystalline at all, but amorphous materials.
LikeLike
A given block of steel can go from one extreme to the other simply with heat treatment. Of course, if the processing adds or subtracts carbon and/or messes with the heat treatment, all bets are off.
Did an intern stint at Inland-Ryerson, when they were still making steel in the USA. My co-workers told me of a ring of EZ-machining steel (leaded 12L14, probably) that had been flame cut and sent to the customer. The shop foreman thought it had been annealed, and was rather upset that his tooling lost its edge in seconds.
(Home machining, I learned that the “D2” on a block of steel from Remnants-r-Us wasn’t random. High speed steel doesn’t fare well against die steel. Machinery’s Handbook became required reading after that…)
LikeLike
I like the pictures. They match what you described when I read The Tally Master (which I highly recommend).
LikeLike
In terms of monetary value, during the Late Bronze Age (2nd Millennium BC), gold, lapis lazuli, and tin were unit-for-unit accorded equal value. That is, you could trade an ounce of tin for an ounce of gold or lapis.
Also, you tend to find arsenical bronze more in the earlier BA, although the hardness of arsenical bronze was prized for things like mirrors and razors.
LikeLike
Tin = gold = lapis lazuli
What a great detail!
LikeLike
Wait, tin and gold are equal in value to Lazarus Long’s opposite gender clones together?
LikeLike
“Lapis Lazuli long” and “Lorelei Lee Long”
LikeLike
Gold and lapis were pretty. But tin was *useful.*
Platinum used to be an annoying impurity that cropped up when refining gold. Nowadays it is a heavily used industrial metal as well as sometimes used on its own for jewelry.
LikeLike
Except for the clever Inca, who figured out if you mixed in the strange unmeltable powder you didn’t need as much gold for your fancy dodads :D They had a way to remove or otherwise make a pure gold outside layer.
LikeLike
Wait, the Incas diluted gold with platinum??? Then again, I think gold was more expensive.
I had a glass professor who told us that platinum nozzles for making fiber glass were rented due to cost; when they wore out you traded them in.
LikeLike
Oops, gold was more expensive 5 +/- years ago.
And if they couldn’t use it any other way sure make your gold go further.
LikeLike
Several years ago (circa 1970), I had a commercially recorded tape (on a 7″ reel, no less) of Rheingold from Der Ring des Nibelungen. The liner notes said they used tin ingots instead of the proper gold for effects in the ransom scene, but the tin was still incredibly expensive, warranting armored car transport.
The tape (and the recorder to play it) is long gone. I have the LP set of the Ring Cycle (same version), but the liner notes are in German. :)
LikeLike
Fascinating information. Thanks for sharing.
LikeLike
Thanks for all the fantastic discussion, guys. That was fun!
And thanks for hosting me, Sarah. You’ve got a great group collected around you. :D
LikeLike
Completely off topic, but- someone was flinging carp:
http://sacramento.cbslocal.com/2017/05/19/oroville-school-covered-in-dead-fish/
LikeLike
Nobody saw them emplaced, supposedly. And nobody noticed a rain of fish. So… teleportation.. only works for carp/fish?
LikeLike
There was a slight targeting error in our testing of the ICBCM. We believe the problem has been fixed.
LikeLike
I’m sure that it’s just a red herring type of tale…
LikeLike
First guess: someone was air-dropping fish for lake stocking and the tank leaked.
Second guess: CA is just strange.
Third thought: the loaves will appear over the weekend. Or all those jokes about the Most High getting Old Testament on California are in the process of coming true and folks had better get ready to have a quick dinner and then relocate.
LikeLike
Surely no one would stock carp, but on the other hand why or how would a waterspout pick up only one species.
LikeLike
Goldfish are a species of carp……
LikeLike
Oh yeah.
LikeLike
It’s California — are there any wildfires in the area? If a water tanker flight hit a carp lake fr its resupply and lost their load en route …
Say, does anyone know if there is factual basis for tales of scuba divers being found dead at wildfire sites?
LikeLike
Today it would be less likely. Seaplanes still use scoops to load the tanks but that would be a relatively shallow draft if I remember. Helos use pumps and hoses. I believe those definitely have guards for brush and objects to prevent damage to pump parts.
LikeLike
There’s one fire (3000 acres on Friday), but way too far south.
Mythbusters did something on the scuba diver; not likely; the biggest sucker hose is a foot in diameter. Some schmuck might be able to climb into a helo’s Bambi Bucket and get himself killed that way, but that’s a pretty elaborate way of suicide.
As best as I recall, the Martin Mars(?) is the big seaplane used for firefighting. I think it uses scoop intake. I’ve seen one on Vancouver Island; a Mariner’s big brother.
Speculation in the national news referred to a waterspout sucking up the fish. Lake Oroville is nearby, but I’m holding out hope for the Mother of All Carpapults.
LikeLike
Because they only needed one school to cover one school?
LikeLike
Someone has an Improbability Generator and this is a (side?) effect thereof?
LikeLike
I spent 35 years in a steel making factory. We made everything from ferrous to nickle based to cobalt based steels, and also nickle based vacuum melted steels for the jet engine industry. It is indeed cheaper to make steel with a higher carbon content and then simply reduce that to the desired level. This is done by first melting the charge, usually for us it was 20k pounds at a time. Then we would transfer the molten steel to a vessel. The process for removing carbon from steel is called Argon Oxygen Decarburization. AOD, for short. From this point on, there is no method to keep the steel hot except for injecting gases into the bottom of the vessel through what are called tuyeres, similar to the old tubes that people used to have to blow through. Oxygen has an affinity to combine with different elements when they are in a molten state, and carbon is one of the first that it combines with. Silicon and aluminum are also very easily to combine with oxygen, and so they tend to be the first elements that are burned off during processing. This produces a chemical reaction known as an aluminothermic reaction, which makes heat. That is what keeps the steel hot. This would be simple, save for the fact that other desireable elements are also combined with oxygen as well. It would be quite cost prohibitive to have to add back chrome, nickle, copper, etc, after you blew the carbon out of a heat. So what is done is that before the heat is brought over to the vessel, a precharge of slag is added to the vessel, consisting of the same material as the lining of the vessel, usually lime and magnesium, or dolomite. During the blow of the carbon, the carbon comes off as carbon monoxide, but the desirable elements are trapped in the slag layer that forms on the top of the bath. Once the carbon has been determined by the lab to be low enough, you enter what is called a reduction process. Simply put, it is similar to the water softener you have in your home. There, you add salt to replace the undesirable deposits such as iron. During steel making, you add silicon and aluminum, the amount based mathematically upon how much oxygen you had to use to reduce the carbon down, and also with an eye to trying to preserve the life of the vessel, sometimes more precharge material is added. Then you stir the heat for a determined amount of time, usually 10 minutes or so, but not with oxygen this time, but with plain argon, which is an inert gas. Some alloys require a certain amount of nitrogen as well, so it is at this point that some of that would be injected as well. From there a full chemical analysis is done, usually withing 10 minutes, using x rays and also a seperate, destructive machine for the carbon and nitrogen check. At this point, corrective additions are made, and, assuming that the temperature is sufficent, the heat is tapped and poured either into ingots, or more often, into a single strand casting machine which at the very end you have round, 18 inch long by 3 inch diameter ingots packed and ready to go, all within about 2 hours. If the temperature is not hot enough, aluminum and silicon is added and oxygen and argon is blown into the bath just until those two elements have been used up, which gains temperature. If the amount of temp needed isn’t too great, another lab check is usually unnecessary, unless the chemistry requirements are extremely tight. I have seen these metals sold to companies making everything from gun parts, to boat propellers, artificial knees and hips, food service equipment, even the spurs on fighting cocks down mexico way. Also we have remelted tail fins from side winder missiles, etc, and so I would suppose that they have their hand in the mix as well. That is the airmelted side of things. There is also another method of steel making, for alloys called super alloys, which are melted under a high vacuum, in order to keep the oxygen and nitrogen out. These are mostly used in jet airplane engines. And the topic of that could be another long drawn out affair, so I will leave it along, unless someone wants to know more.
LikeLike
Dude! Two words of advice: paragraph breaks.
It doesn’t matter how fascinating the content if eyes start glazing over in the face of a wall of text.
LikeLike
Er, agreed on the paragraph breaks (I’m guilty of that too), but this is fascinating.
I dealt with construction steel (a lot of certified A-36 went into nuclear plants in the midwest; we did a fair share), but I never learned the details of the more exotic stuff. The Basic Oxygen furnace was the ticket for structural steel, circa 1972. I don’t know how Chinese Mystery Metal is made; not sure I want to.
LikeLike
From what I’ve read of Chinese (and Japanese) folk-lore, I don’t either.
Although there’s a really spooky US tale about an iron-foundry owner and his pack of dogs… https://hsp.org/blogs/hidden-histories/the-legend-or-haunting-of-the-hounds-of-colebrook-furnace-lebanon-county-pa
LikeLike
My apologies. I wrote this quickly, trying to stuff too much into it, and so didn’t take the time to do as neat a job as I should have. Your correction to me is appreciated, not taken as being critical.
LikeLike
A number of years back, here in the third world, we had a crew pouring cement, and the starter rope’s rebound spring on the mixer broke, and badly.
So, I stripped the spring from a busted lawnmower… and it was too big. Using the gas stove in my house, I annealed, cut, bent, tempered, and did a quick & dirty heat treat to resize the spring, and it was still too big. Rinse, repeat, and got the machine running within a half hour of the breakdown.
LikeLike
Holy crap. I wouldn’t have thought you could do all that heating and quenching and cooling in 30 minutes, let alone twice.
LikeLike
I used to work with a young American Indian man from Michigan’s upper peninsula in the machine shop. He said the elders in his tribe had copper knives that were as good as steel, but it was a secret how to make them reserved for a few older men. They wouldn’t tell him how, saying he would run as fast as he could and sell it to the white people for a few bucks. They were right, he was a first class jackass to work with and mean. I came to work wearing new boots and he immediately ran up and stomped on them to mark them up. The kind of guy who keys a Mercedes in the parking lot because nobody should have something he doesn’t.
LikeLike
In which case I’d want to *see* one of those knives. Jerk like that would lie just to keep your attention.
LikeLike
British show has comedy bit on changing from stone to bronze tools. https://m.youtube.com/watch?v=-EGAtLGDU7M
LikeLike
In “The Doomsday Myth: 10,000 Years Of Economic Crises”, Charles Maurice & Charles W. Smithson propose that the Greeks began using iron instead of bronze because the emergence of the Sea Peoples cut Greece off from receiving overseas tin.
As the price of tin increased, iron became relatively less expensive to make. And once they began making iron, Greek smiths became more proficient and efficient at using this “new” metal.
LikeLike